Jacob Williamson-Rea
Table of Contents
An Examination of Carboniferous Arthropods
Introduction
The most successful animals on the planet, Phylum Arthropoda encompasses a vast variety of highly successful organisms. The name Arthropoda is derived from the Greek arthos meaning “joint,” and podos meaning “foot.” When examining ecological variety, it can be clearly determined that arthropods are one of the most successful and versatile organisms on the planet. The fossil record indicates that the arthropods first appeared at the beginning of the Cambrian Period, approximately 540 million years ago. Various arthropod species occupy numerous niches; arthropods’ segmented body plans enable various species to inhabit almost any environment.
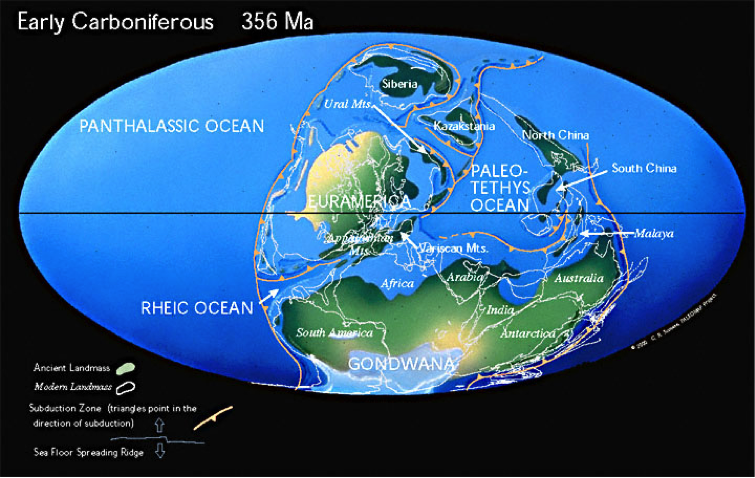
Below are recreations of the world’s appearance during the Late and Early Carboniferous Period:
During the Carboniferous Period, from 359-299 million years ago, a spike in atmospheric oxygen levels led to an increase arthropod size. Compared to today’s 21%, oxygen constituted about 35% of the Carboniferous atmosphere. The Carboniferous millipede reached 10 feet in length; gigantism was normal for all organisms during this time period (Beerling). The figure below indicates the drastic spike during the Early Carboniferous (Harrison).

Solid line based on various approaches including the balance of sedimentary rock abundance (Berner & Canfield 1989), dotted line based on carbon and sulphur isotopic changes, and incorporating feedbacks from atmospheric oxygen levels on biogeochemical cycling (Berner 2006), dashed line (Bergman et al. 2004).
Phylum Arthropoda is grouped into five subphylums: Trilobitomporpha, Chelicerata, Crustacea, Insecta, and Myriapoda.
- Subphylum Trilobitomorpha includes the well-known and most commonly fossilized trilobite, which is now extinct.
- Subphylum Chelicerata encompasses arachnids, horseshoe crabs, and eurypterids.
- Subphylum Crustacea covers lobsters, crabs, shrimp, crayfish, barnacles, prawns, krill, and ostracodes.
- Subphylum Insecta is the largest and most diverse group of arthropods.
- Subphylum Myriapoda is defined by the presence of biramous appendages.
Although arthropods often have poor preservation potential due to their organic chitinous exoskeletons, the trilobite’s calcified exoskeleton makes it an excellent group to examine evolutionary history. However, this means significant bias exists in the Arthropoda fossil record. With recognition of this bias, a section of this paper will analyze the phylum’s phylogeny and incorporate the arthropods found in various Lagerstätten. The Burgess Shale, a Lagersätte that contains preserved nonmineralized tissues, provides well-preserved arthropod specimens and a rare glimpse into the otherwise unpreserved lives of arthropods such as Marella, Olenoides, and Naraoia. Along with The Burgess Shale, several other Lagerstätte contain important arthropod finds: Chengjian Lagerstätte, Sirius Passet, Emu Bay Shale, and Fezouata arthropods will be examined.
The following segments contain an examination of various aspects of Phylum Arthropoda during the Carboniferous period, as well as an analysis of Pennsylvanian Carboniferous Arthropods. A background detailing the planet’s condition during this time period will provide context for the more specific subjects discussed. Arthropoda evolutionary history and molecular phylogeny will provide a historic background in order to formulate the basis of the phylum. Arthropoda’s fossil record will be scrutinized with regard to possible bias in the record. Various aspects of anatomy in terms of fossil identification will differentiate between subphylums, classes, families, and orders.
The following segments contain an examination of various aspects of Phylum Arthropoda during the Carboniferous period, as well as an analysis of Pennsylvanian Carboniferous Arthropods. A background detailing the planet’s condition during this time period will provide context for the more specific subjects discussed. Arthropoda evolutionary history and molecular phylogeny will provide a historic background in order to formulate the basis of the phylum. Arthropoda’s fossil record will be scrutinized with regard to possible bias in the record. Various aspects of anatomy in terms of fossil identification will differentiate between subphylums, classes, families, and orders.
Evolutionary History
Arthropods first evolved as early as 540 million years ago in the Cambrian Period. Small arthropods have been found in China’s Early Cambrian fossil beds, but some paleontologists propose that Ediacaran specimens such as Spriggina were arthropods. Arthropods initially live strictly in marine habitats, but would later become the first animals to colonize the land’s surface (Briggs). Although still debated by some, recent evidence suggests that Phylum Arthropoda is a monophyletic group – meaning that all arthropods are descended from a common ancestor (Edgecombe). Earlier studies, stating that the phylum was polyphyletic, were based incorrectly on the idea that an overwhelming morphological disparity must have more than one single common origin. More recently analyses determine that the vast variances of arthropods all stem from a shared, common origin.
All of the arthropod subphyla, although quite varied, display segmented bodies and jointed appendages. Subphyla Trilobitomorpha, Chelicerata, Crustacea, Hexapoda (Insects), and Myriapoda are all related evolutionarily. Studies conducted by Omar Rota-Stabelli and others use molecular data combined with a variety of anatomical and developmental characteristics. Their findings produced a phylogeny quite similar to arrangements based on embryological and morphological studies (Prothero). Rota-Stabelli et. al determined that the chelicerates are monophyletic and separate from the other arthropods. Two groups are part of Arachnata: Trilobita and Chelicerata. Three major groups make up the Mandibulata clade: Crustacea, Myriapoda, and Insecta.
Based on early Lagerstätten evidence, paleontologists consider a polyphyletic origin of the arthropods improbable (Briggs). Specimens from early Cambrian, when arthropods first evolved and rapidly diversified, explain the bridge between major arthropod groups.
The extinct trilobites’ calcified exoskeleton allows one to study their ontogeny. The earliest, or protaspid stage, is when the juvenile has one, basic, circular plate. Then, the thoracic segments began to appear, separating the pygidium from the cephalon – the merasipid stage. This form tends to vary, but eventually the organism reaches the holapsid stage, which is when all of the adulet thoracic segments are present. Most trilobites, upon reaching the holapsid stage, would have to be benthic detritus feeders because of their size.
After the Devonian extinction, only one order of Subphylum Trilobitomorpha remained: Proetida, a long-lived and conservative group of trilobites, survived the Late Devonian Extinction. They were rare during the Carboniferous period, and went extinct during the Late Permian Period. Proetids, relatively smaller trilobites, displayed tubercles and pits on their exoskeletons.
The extinct trilobites’ calcified exoskeleton allows one to study their ontogeny. The earliest, or protaspid stage, is when the juvenile has one, basic, circular plate. Then, the thoracic segments began to appear, separating the pygidium from the cephalon – the merasipid stage. This form tends to vary, but eventually the organism reaches the holapsid stage, which is when all of the adulet thoracic segments are present. Most trilobites, upon reaching the holapsid stage, would have to be benthic detritus feeders because of their size.
After the Devonian extinction, only one order of Subphylum Trilobitomorpha remained: Proetida, a long-lived and conservative group of trilobites, survived the Late Devonian Extinction. They were rare during the Carboniferous period, and went extinct during the Late Permian Period. Proetids, relatively smaller trilobites, displayed tubercles and pits on their exoskeletons.
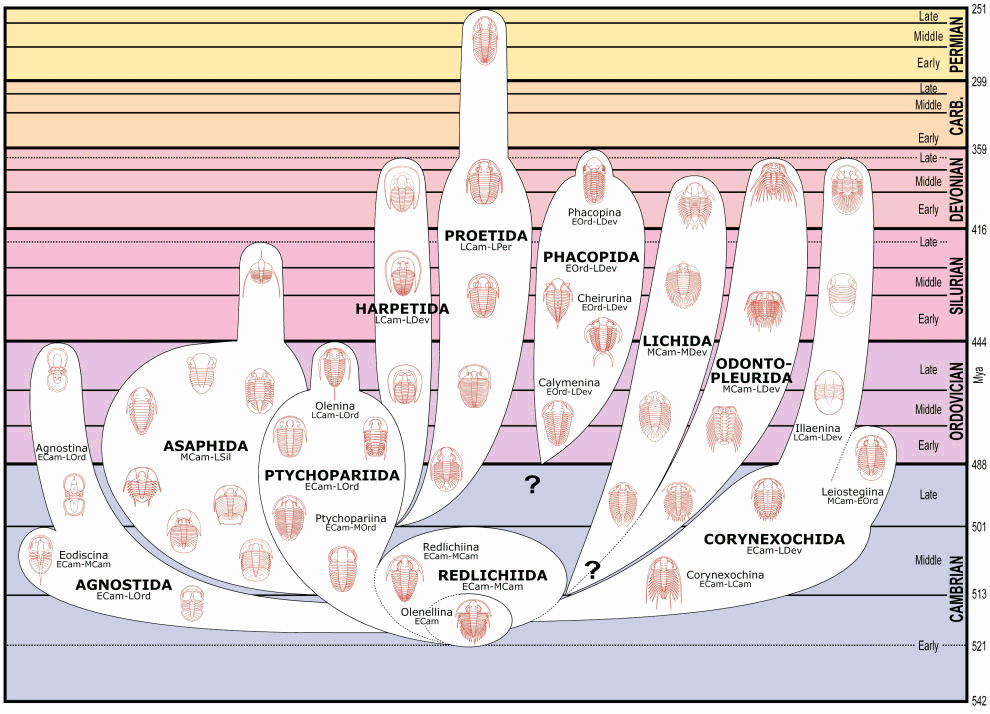
Trilobites were most diverse during the Cambrian, and this visual provides context for the difference in trilobite diversity between the Middle Cambrian and Late Devonian/Early Carboniferous.
Fossil Record
Due to arthropods’ chitonous exoskeletons, the preservation potential of these organisms is often poor. However, the trilobite’s calcified exoskeleton leads to a greater representation of trilobites in the fossil record; this may have initially caused bias in terms of trilobite diversification and abundance. However, trilobites numbers were scarce and dwindling in the Pennsylvania area during the Carboniferous Period. Arthropods with chitinous exoskeletons have been preserved in Lagerstätten and amber, but trilobites are quite commonly preserved in limestone and shale. In 2012, a juvenile polymerid trilobite with non-biomineralised tissues found in the Orsten Konservat-Lagerstätte revealed information about trilobite interior anatomy by using Synchroton-readiation X-ray tomographic microscopy. (http://journals.plos.org/plosone/article?id=10.1371/journal.pone.0035625)
Fossil trackways, a trace fossil of an organism’s steps, also reveal information about trilobites in the Carboniferous Period. The largest arthropod trackway was found in Elk County, Pennsylvania. Displayed in the Carnegie Museum of Natural History, the trackway is hypothesized to be from a eurypterid.
Fossil trackways, a trace fossil of an organism’s steps, also reveal information about trilobites in the Carboniferous Period. The largest arthropod trackway was found in Elk County, Pennsylvania. Displayed in the Carnegie Museum of Natural History, the trackway is hypothesized to be from a eurypterid.
Fossil Identification Guide
Subphylum Trilobitomorpha
Three Lobes moving laterally propel the trilobite forward.
Segments were fused into 3 components moving anterior-posterior.
Segments were fused into 3 components moving anterior-posterior.
Primary Parts of the Trilobite:
Subphylum Chelicerata
No antennae
4 pairs of pedipolp and chelicera
Cephalothorax and abdomen
4 pairs of pedipolp and chelicera
Cephalothorax and abdomen
Subphylum Crustacea
2 pairs of antennae
5 pairs of legs/attachments
Cephalothorax and abdomen
5 pairs of legs/attachments
Cephalothorax and abdomen
Subphylum Hexapoda (Insecta)
1 pair of antennae
3 pairs of legs/attachments
Head, 3 segmented thorax, segmented abdomen, wings, and appendages.
3 pairs of legs/attachments
Head, 3 segmented thorax, segmented abdomen, wings, and appendages.
Subphylum Maryiopoda
1 pair of antennae
Variable number of legs and attachments - often many
Head and segments
Variable number of legs and attachments - often many
Head and segments
Pennsylvania and Appalachian Basin Fossil Guide
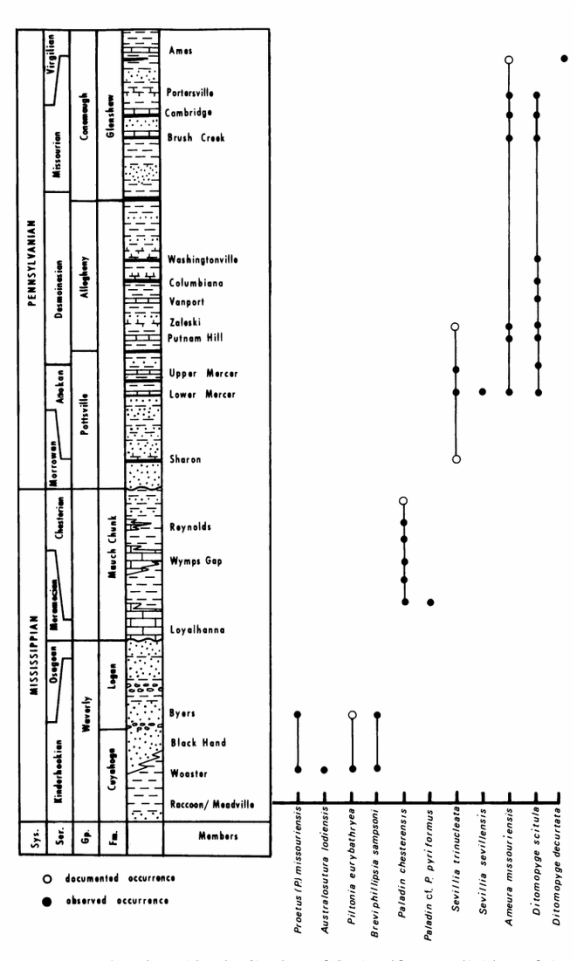
Pennsylvania forms of trilobites from the Carboniferous Period are poorly known, so fossil evidence from north-central Appalachian Basin strata has been used to examine and describe these poorly known forms. The Appalachian Basin includes eastern Ohio, western Pennsylvania, northern West Virginia, and western Maryland. The below figure illustrates the presence of particular trilobites within Appalachian Basin strata.
1. Waverly Group - Cuyahoga and Logan Formations (Mississippian): Although the distribution is sporadic, these two formations contain the best known trilobite faunas (Brezinski). The following trilobites have been found throughout the sparse outcrops.
Found in the Cuyahog and Logan Formations:
Found in the Cuyahog and Logan Formations:
|
Proetus (Pudoproetus) missouriensis: An abundance of this species has been found in the Byers Sandstone (Shumard). The Bryers Sandstone was deposited in a shallow shelf setting. Pudoproetus species can be differentiated by their inflated lateral occipital lobes and ten thoracic segments. This species survived the Late Devonian extinction thanks to their adaptation to shallow waters (Feist). |
|
Australosutura lodiensis: A rare fauna found in Sciotoville, Wooster, and Meadville Shales, Australosutura lodiensis also survived the Late Devonian extinction, but fewer specimens have been uncovered. (Meek) Characterized by:
|
|
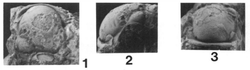
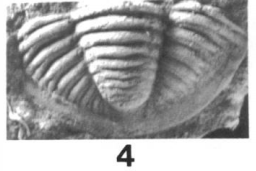
7-12, Piltonia eurybathrea (Hessler). 7, 8, 12, dorsal, lateral, and anterior views of incomplete unexfoliated holotype cranidium, OSU 22805, x 2.5; 9-11, internal mold of pygidium, OSU 22532, x 2.0. (Brezinski)
|
Piltonia eurybathrea: Piltonia eurybathrea, along with the following Breviphillipsia sampsoni, are found within the Meadville and Wooster shales, but have also been found in the Bryers Sandstone (Hessler). The characteristic exterior bumps, or ornamentation, provided extra rigidity and protection. See below specimen, Breviphillipsia sampsoni for comparisons. |
|
Breviphillipsia sampsoni: Breviphillipsia sampsoni fossils are also found within the Meadville and Wooster shales, and have been discovered in the Bryers Sandstone. (Vodges) Breviphillipsia samsoni appears quite similar to the above Piltonia Eurybathrea, but the following can be used to differentiate between the two.
|
2. Mauch Chunk - Greenbrier Formation (Mississippian): Comprised of siltstone, shale, sandstone, and conglomerate The Mauch Chunk Greenbrier Formation was originally deposited on a large basin, with sediment from various highlands. Limestone deposition indicates the fluctuation of sea levels. The following specimens have been discovered in the Greenbrier Formation.
Found in the Greenbrier Formation:
Found in the Greenbrier Formation:
|
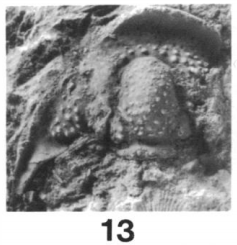
28, 29, 31, 32, 34 Paladin chesterensis. 28, 29, 31, dorsal, posterior, and lateral views of unexfoliated pygidium, CMNH 34606, x 3.0; 32, 34, dorsal, anterior, and lateral views of partially exfoliated cranidium, CMNH 34608. (Brezinski)
|
Paladin chesterensis:
This species has been found at various Mauch Chunk intervals as well as specific Greenbrier Group carbonate facies. Paladin chesterensis can be identified by its:
|
Also found in the Mauch Chunk Formation:
 Scoyenia ichnoguilds
Scoyenia ichnoguilds
The high number of Scoyenia ichnoguilds, invertebrate trace fossil burrows, in the Mauch Chunk indicates “a terrestrial invertebrate fauna dominated by both a mobile epifauna and a shallow, deposit feeding fauna” (Fillmore 35). Along with Scovenia ichnoguilds, Taenidium worm burrows can be found in the Mauch Chunk Formation.
3. Pottsville Group (Pennsylvanian): The Pottsville Group, comprised of mostly sandstone and conglomerate, was originally a non-marine depositional environment. The Pottsville contains the following trilobite species.
|
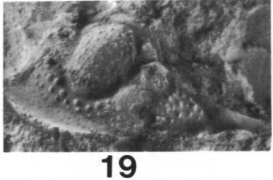
3, 15, 17, Sevillia trinucleata. 1-3, dorsal, anterior, and lateral views of partial, unexfoliated paratype cranidium, OSU 28135a, x2.0; 15, latex cast of external mold of paratype pygidium, OSU 28135b, x2.0; 17, dorsal view of partially exfoliated pygidium, OSU 29174, x 2.0. (Brezinksi)
|
Sevillia trinucleata: Sevillia t. can be identified by its:
|
Although extremely rare, the following trilobites can also be found in The Pottsville Group: Sevillia sevillensis,Ditomopyge scitula,and Ameura missouriensis (Shumard).
4. The Conemaugh Group (Pennsylvanian):
 Casselman Formation Outcrop
Casselman Formation Outcrop
The Casselman Formation (Pennsylvanian):The Casselman Formation constitutes the upper part of the Conemaugh Group, and the Glenshaw Formation makes up the lower half. This group consists of shale, sandstone, siltstone, thin limestone, red beds, and coal. The Casselman is comprised of almost entirely fresh water rocks.
The Casselman Formation (Pennsylvanian):The Casselman Formation constitutes the upper part of the Conemaugh Group, and the Glenshaw Formation makes up the lower half. This group consists of shale, sandstone, siltstone, thin limestone, red beds, and coal. The Casselman is comprised of almost entirely fresh water rocks.
Found in the Casselman Formation:
|
|
Ameura missouriensis - Ameura m. can be differentiated by its well-developed cephalon (head), as well as its long pygidium that contains central axial lobe segments. |
The Glenshaw Formation (Pennsylvanian):
The Glenshaw Formation is the lower portion of the Conemaugh Group.
|
14, 16, 18-20, 22, 26, 27, Ditomopyge scitula. 14, 16, dorsal and oblique views of unexfoliated incomplete cephalon, OSU 29170, x 3.0; 18, 26, dorsal and lateral views of crushed exoskeleton, CMNH 34612, x 3.5; 19, 20, 27, dorsal, posterior, and lateral views of unexfoliated pygidium, CMNH 34617, x 4.0; 22, dorsal view of unexfoliated cranidium, CMNH 34616, x 4.0. 2
|
Ditomopyge scitula: By far the most common Pennsylvanian trilobite found in The Appalachian Basin's Conemaugh Group. Characterized by:
|
Much less common than Ditomopyge scitula, these trilobites can be found within the Conemaugh Group's Casselman and Glenshaw Formations: Ditomopyge scitula and Amerua missouriensis.
5. Llewellyn Formation (Mid-Late Pennsylvanian): The Llewllyn Formation, located in St. Clair, Pennsylvania, contains dark, repetitive sequences of sandstone, shale, siltstone, anthracite coal, and conglomerate. The Llewellyn Formation was deposited on a flat plain. Changes in sea level, along with the shifting tendencies of the highlands and rivers, enabled dense forests to prosper on this plain. This organic matter would later be buried and turned to coal. (Edmunds)
Found in the Llewellyn Formation:
Found in the Llewellyn Formation:
Diplichnites gouldi is an invertebrate trace fossil. The appendage imprints can clearly be defined; these are interpreted as likely locomotion traces. The trackway displays the four Arthropoda required digit appendage patterns:
- Curvatures suggesting angular changes in locomotion
- Internal and external widths sequenced in alternating, staggered, and symmetrical patterns.
- Identifiable, measurable stride distance.
- Morphology of appendage and digit traces in a V-shaped position to the angle of mid-line symmetry. (Smith)
Notable Arthropod Locations and Specimens Outside of Pennsylvania and Appalachian Basin strata:
Chouteau Formation - Saline County, Missouri (Lower Mississippian):
 Comptonaspis swallowi from Kinderhookian Stage
Comptonaspis swallowi from Kinderhookian Stage
Comtonaspis swallowi, a member of the order Protetida, is a rare species distinguished by its smooth exoskeleton.
Edwardsville Formation - Crawfordsville, Indiana:
Pallero Member, Bajo de Véliz Formation - San Luis Province, Argentina:
“The nearly complete and well preserved fossil is in a single dark gray slaty shaly flag, and shows a considerable amount of morphological and structural details. Most of the legs are visible over the surface of the flag, but some were located underneath with the help of X-Ray analysis.” (Hünicken)
Although sparse and often rare, Appalachian Basin and Pennsylvania trilobites can be found and examined, mostly thanks to the work of David K. Brezinski.
References Cited
Beerling, David. Emerald Planet: How Plants Changed Earth’s History. New York: Oxford,
2007. Print.
Brezinski, David K. “Appalachian Carboniferous Trilobites.” Journal of Paleontology 62.6
(Nov. 1988): 934-945.
Brezinski, David K., and James H. Stitt. “Ditomopyge Scitula (Meek and Worthen) from the Lower Pennsylvanian of Central Missouri and Central Texas." Journal of Paleontology 56.5 (1982): 1242–1250.
Briggs, Derek E.G. “Arthropod Paleobiology.” Paleontological Society 11.4 (Fall 1985): 361-
367.
Briggs, Derek E.G., and Richard A. Fortey. “The Early Radiation and Relationships Among
Major Arhtropod Groups.” Science 246.4927 (1989): 241-243.
Briggs, Derek E.G. and W.D. Ian Rolfe. “A Giant Arthropod Trackway from the Lower
Mississipian of Pennsylvania.” Journal of Paleontology 57.2 (1983): 377-390.
Brusca, Richard C. “Unraveling the History of Arthropod Biodiversification.” Annals of the
Missouri Botanical Garden 87.1 (Winter, 2000): 13-25.
Edgecombe, Gregory D., and David A. Legg. “The Arthropod Fossil Record.” Arthropod
Biology and Evolution: Molecules, Development, Morphology. Eds. Boxshall, Geoffrey,
Giuseppe Fusco, and Alessandro Minelli. New York: Springer, 2013. 393-417.
Edmunds, W.E “Coal in Pennsylvannia.” Pennsylvania Geological Survey, Education Series 7.
(2002): Print.
Eriksson, Mats E. and Fredrik Terfelt. “Exceptionally Preserved Cambrian Trilobite
Digestive System Revealed in 3D by Synchroton-Radiation X-Ray Tomographic Microscopy.” PLOS. 25 April, 2012.
Fillmore, David L., Spencer G. Lucas, and Edward L. Simpson. Ichnology of the Mississippian
Mauch Chink Formation, eastern Pennsylvania. New Mexico Museum of Natural
History and Science: 2012. 35-37. Print.
Feist, Raimund, and Morris S. Petersen (1995). Origin and spread of Pudoproetus, a survivor of the Late Devonian trilobite crisis. Journal of Paleontology, 69, pp 99-109.
Harrison, Jon F, Alexander Kaiser, and John M. VandenBrooks. “Atmospheric oxygen level
and the evolution of insect body size.” Proceeding of the Royal Society B: Biological
Sciences. 26 May 2010.
< http://rspb.royalsocietypublishing.org/content/277/1690/1937.short>
Hessler, R. R. 1962. Secondary segmentation in the thorax of trilo- bites. Journal of Paleontology, 36:1305-1312.
Hünicken, Mario A. “A Giant Fossil Spider (Megarachne servinei) from Bajo de Veliz, Upper
Carboniferous, Argentina.” Boletin de la Academia Nacional de Ciencas. 53: 1-14.
1980.
Lofgren, Andrea Stockmeyer, Roy E. Plotnick and Peter J. Wagner. “Morphological Diversity
of Carboniferous Arthropods and Insights on Disparity Patterns through the Phanerozoic.” Paleobiology 29.3 (Summer, 2003): 349-368.
Meek, F. B. 1875. A report on some invertebrate fossils of the Waverly Group and Coal Measures of Ohio, p. 268-347. In J. S. Newbury (ed.), Report of the Geological Survey of Ohio, Vol. 2. Geology and Paleontology, Pt. 2. Columbus, Ohio.
Scotese, Christoper R. “Paleomap Project.” Paleomap Project. Accessed 21 Feb. 2016
< http://www.scotese.com/Default.htm>
SHUMARD, B. F. 1855. Description of a geological section on the Mississippi River from St. Louis to Commerce. Geological Survey of Missouri 1st and 2nd Annual Report, Pt. 2, p. 185-208.
Smith, Casey J. “The First Reported Arthropod Trackways from the Pennsylvanian
Llewellyn Formation.” Geological Society of America Abstracts with Programs Vol. 41, No. 3, p. 88
Smithsonian Museum of Natural History. “Burgess Shale Fossil Specimens.” Smithsonian
Institution. Accessed 20 Feb. 2016
<http://paleobiology.si.edu/burgess/burgessSpecimens.html>
Vodges, A. W. 1887. The genera and species of North American trilobites. Annals of the New York Academy of Sciences, 4:69-105
2007. Print.
Brezinski, David K. “Appalachian Carboniferous Trilobites.” Journal of Paleontology 62.6
(Nov. 1988): 934-945.
Brezinski, David K., and James H. Stitt. “Ditomopyge Scitula (Meek and Worthen) from the Lower Pennsylvanian of Central Missouri and Central Texas." Journal of Paleontology 56.5 (1982): 1242–1250.
Briggs, Derek E.G. “Arthropod Paleobiology.” Paleontological Society 11.4 (Fall 1985): 361-
367.
Briggs, Derek E.G., and Richard A. Fortey. “The Early Radiation and Relationships Among
Major Arhtropod Groups.” Science 246.4927 (1989): 241-243.
Briggs, Derek E.G. and W.D. Ian Rolfe. “A Giant Arthropod Trackway from the Lower
Mississipian of Pennsylvania.” Journal of Paleontology 57.2 (1983): 377-390.
Brusca, Richard C. “Unraveling the History of Arthropod Biodiversification.” Annals of the
Missouri Botanical Garden 87.1 (Winter, 2000): 13-25.
Edgecombe, Gregory D., and David A. Legg. “The Arthropod Fossil Record.” Arthropod
Biology and Evolution: Molecules, Development, Morphology. Eds. Boxshall, Geoffrey,
Giuseppe Fusco, and Alessandro Minelli. New York: Springer, 2013. 393-417.
Edmunds, W.E “Coal in Pennsylvannia.” Pennsylvania Geological Survey, Education Series 7.
(2002): Print.
Eriksson, Mats E. and Fredrik Terfelt. “Exceptionally Preserved Cambrian Trilobite
Digestive System Revealed in 3D by Synchroton-Radiation X-Ray Tomographic Microscopy.” PLOS. 25 April, 2012.
Fillmore, David L., Spencer G. Lucas, and Edward L. Simpson. Ichnology of the Mississippian
Mauch Chink Formation, eastern Pennsylvania. New Mexico Museum of Natural
History and Science: 2012. 35-37. Print.
Feist, Raimund, and Morris S. Petersen (1995). Origin and spread of Pudoproetus, a survivor of the Late Devonian trilobite crisis. Journal of Paleontology, 69, pp 99-109.
Harrison, Jon F, Alexander Kaiser, and John M. VandenBrooks. “Atmospheric oxygen level
and the evolution of insect body size.” Proceeding of the Royal Society B: Biological
Sciences. 26 May 2010.
< http://rspb.royalsocietypublishing.org/content/277/1690/1937.short>
Hessler, R. R. 1962. Secondary segmentation in the thorax of trilo- bites. Journal of Paleontology, 36:1305-1312.
Hünicken, Mario A. “A Giant Fossil Spider (Megarachne servinei) from Bajo de Veliz, Upper
Carboniferous, Argentina.” Boletin de la Academia Nacional de Ciencas. 53: 1-14.
1980.
Lofgren, Andrea Stockmeyer, Roy E. Plotnick and Peter J. Wagner. “Morphological Diversity
of Carboniferous Arthropods and Insights on Disparity Patterns through the Phanerozoic.” Paleobiology 29.3 (Summer, 2003): 349-368.
Meek, F. B. 1875. A report on some invertebrate fossils of the Waverly Group and Coal Measures of Ohio, p. 268-347. In J. S. Newbury (ed.), Report of the Geological Survey of Ohio, Vol. 2. Geology and Paleontology, Pt. 2. Columbus, Ohio.
Scotese, Christoper R. “Paleomap Project.” Paleomap Project. Accessed 21 Feb. 2016
< http://www.scotese.com/Default.htm>
SHUMARD, B. F. 1855. Description of a geological section on the Mississippi River from St. Louis to Commerce. Geological Survey of Missouri 1st and 2nd Annual Report, Pt. 2, p. 185-208.
Smith, Casey J. “The First Reported Arthropod Trackways from the Pennsylvanian
Llewellyn Formation.” Geological Society of America Abstracts with Programs Vol. 41, No. 3, p. 88
Smithsonian Museum of Natural History. “Burgess Shale Fossil Specimens.” Smithsonian
Institution. Accessed 20 Feb. 2016
<http://paleobiology.si.edu/burgess/burgessSpecimens.html>
Vodges, A. W. 1887. The genera and species of North American trilobites. Annals of the New York Academy of Sciences, 4:69-105