Carboniferous Paleobotany: Lycophytes & SphenophytesTyler Norris
|
Introduction:
Although often overlooked in fossil study due to an animal-centric viewpoint, ancient plants provided the necessary terrestrial step life needed to be successful out of marine environments. Emerging during the Silurian Period, the vascular plants (tracheophytes) reigned during the Carboniferous (Figure 9). The lycophytes and the sphenophytes are two major groups of vascular plants that reproduce via spores and dominated the wet and low-lying swamps of the Carboniferous. Lycophytes (such as Lepidodendron and Sigillaria) and the sphenophytes (such as Calamites) are common fossils collected in Pennsylvania. Much of the coal used by humans, especially in PA, is derived from the remains of the extinct varieties of lycophytes and sphenophytes. These plants have extant relatives that are seen today covering moist soils, but are small and herbaceous.
Although often overlooked in fossil study due to an animal-centric viewpoint, ancient plants provided the necessary terrestrial step life needed to be successful out of marine environments. Emerging during the Silurian Period, the vascular plants (tracheophytes) reigned during the Carboniferous (Figure 9). The lycophytes and the sphenophytes are two major groups of vascular plants that reproduce via spores and dominated the wet and low-lying swamps of the Carboniferous. Lycophytes (such as Lepidodendron and Sigillaria) and the sphenophytes (such as Calamites) are common fossils collected in Pennsylvania. Much of the coal used by humans, especially in PA, is derived from the remains of the extinct varieties of lycophytes and sphenophytes. These plants have extant relatives that are seen today covering moist soils, but are small and herbaceous.
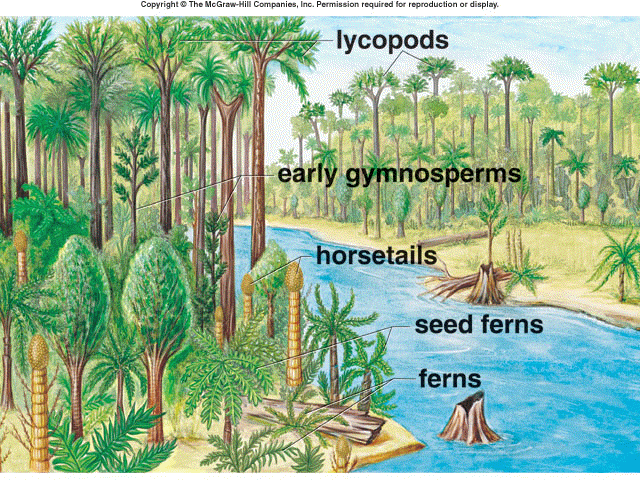
The Carboniferous swamps were ideal for the formation of coal (Figure 1). The swamps extracted much of the carbon dioxide out of the late Paleozoic atmosphere, effectively transitioning the period’s greenhouse conditions to “icehouse” effects by the end of the Carboniferous (Prothero, 2013). Burning coal today releases carbon dioxide stored since that time. The Carboniferous swamps gave a home to amphibians and other newly evolved tetrapods. The only predators to the Carboniferous flora were arthropods, which were also quite large and successful during that time.
Lycophytes, also known as club mosses or quill warts (although being neither moss nor wart), were a major constituent of Devonian forests, which is a time when vascular plants began to prosper. This group is split into the large woody trees (now extinct) and the small, woodless varieties that occasionally blanket moist forests today (Figure 2). Early Lycophytes, such as the low-growing plant Barawanathia, appear in the late Silurian. Thick wooded late Paleozoic lycophytes, such as Lepidodendron and Sigillaria, towered to heights of 120 ft. An extant species of lycophyte is the “ground pine” Lycopium, a small and herbaceous plant that lives in moist forested areas such as modern Pennsylvania. These plants do not have as much woody tissue as their extinct relatives, but have similar roots and leaf-life structures (Prothero, 2013). Lycophytes today are sometimes are seen in holiday decorations in garland and were even used in fireworks (Cobb, 2005).
Figure 2. (Left) The modern Lycophyte Lycopium, (a.k.a. "ground pine") surrounded by leaf litter. Photo taken by author at the University of Pittsburgh at Johnstown campus in a wet, lowland floodplain area.
(Right) An artist’s rendition of a typical Carboniferous Lepidodendron tree with some close-up anatomical features. (http://mtbstravaiger73.blogspot.com/2014_03_01_archive.html)
(Right) An artist’s rendition of a typical Carboniferous Lepidodendron tree with some close-up anatomical features. (http://mtbstravaiger73.blogspot.com/2014_03_01_archive.html)
Sphenophytes, also known as horsetails or scouring rushes, are another tracheophyte group that appears alongside the lycophytes, especially in Carboniferous swamps. All Sphenophytes have long and hollow stems which have splayed whorls of radiating leaves that are attached to joints. Such horsetails, primarily Calamites, grew in stems over 60 ft tall. Recent studies suggest that Calamites may have been closely related to an extant sphenophyte species of Equisetum (Grauvogel-Stamm, 2009). These modern horsetails are much smaller and can be seen in sandy and gravelly soils close to rivers, streams, and swamps (Figure 3). They have fibrous stems composed of abrasive silica and were used by pioneers as scouring pots. Additionally, the unique incremental pattern at which the horsetail shoots branch off potentially inspired mathematician John Napier to investigate logarithmic trends (Sacks, 2011).
Figure 3. (Left) A modern Sphenophyte belonging to the genus Equisetum (the only extant genus) (flickr photo, http://whyy.org/cms/youbetyourgarden/an-ancient-weed-known-as-horsetail/).
(Right) An artist’s depiction of a Calamites covered Carboniferous landscape (Walter Myers, http://www.arcadiastreet.com).
(Right) An artist’s depiction of a Calamites covered Carboniferous landscape (Walter Myers, http://www.arcadiastreet.com).
By the end of the Carboniferous, ferns and early gymnosperms began to dominate the landscape and eventually replaced the lycophytes and sphenophytes, pushing them into the remaining wet lowlands. Fossil studies prove that the abundances of lycophytes in some regions drastically decreased towards the end of the Carboniferous due to the drier conditions of the Permian period (Phillips, 1980). The arid environment of the Permian was due to the slow collision of the ancient continents Gondwana and Euramerica, which eventually formed the supercontinent Pangea. The tree-sized Sphenophytes disappeared by the end of the Carboniferous, but smaller forms persisted through the Permian and Triassic. Today, however, roughly 15 species of the horsetail genus Equisetum remain (Taylor et al, 2009).
Evolutionary History & Fossil Record
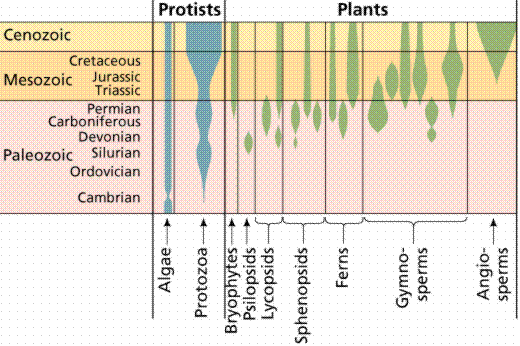
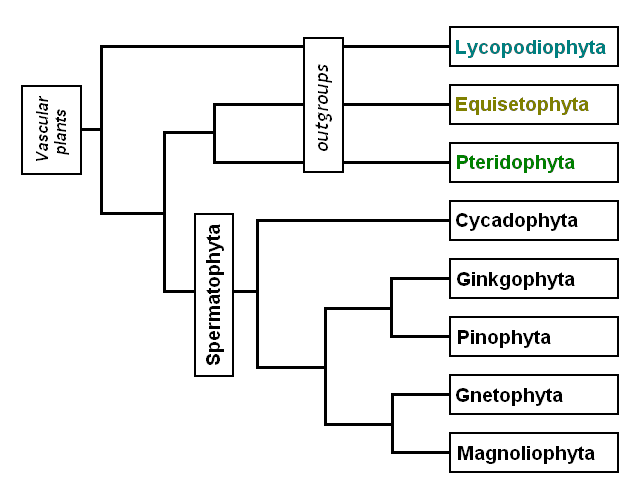
Believed to originate from cyanobacteria, terrestrial land plants arose in the Late Ordovician period (Stewart & Rothwell, 1993). The Kingdom Plantae radiated in the late Silurian Period as it began to colonize terrestrial niches. The earliest terrestrial plants were mosses (Bryophyta) that were able to shut down their metabolism in order to survive harsh conditions, such as periods of drought or frost (Prothero, 2013). Green plants began to split into two divisions: ones tolerant of seawater (chlorophytes) and those that grow near freshwater (charophytes). The charophyta are ancestral to the embryophytes, which branch into vascular and nonvascular varieties. The vascular tracheophytes contain the spore producing lycophytes and sphenophytes, along with pteridophytes and progymnosperms (Figure 4 & 5).
Believed to originate from cyanobacteria, terrestrial land plants arose in the Late Ordovician period (Stewart & Rothwell, 1993). The Kingdom Plantae radiated in the late Silurian Period as it began to colonize terrestrial niches. The earliest terrestrial plants were mosses (Bryophyta) that were able to shut down their metabolism in order to survive harsh conditions, such as periods of drought or frost (Prothero, 2013). Green plants began to split into two divisions: ones tolerant of seawater (chlorophytes) and those that grow near freshwater (charophytes). The charophyta are ancestral to the embryophytes, which branch into vascular and nonvascular varieties. The vascular tracheophytes contain the spore producing lycophytes and sphenophytes, along with pteridophytes and progymnosperms (Figure 4 & 5).
Ancestral examples of vascular land plants appear in the late Silurian and Devonian periods. These include the transitional rhyniophytes Cooksonia and Rhynia. Such vascular plants developed spore bearing forms of reproduction. Interestingly, spores from the fossil record are found 50 Myr before the first known plant fossils appeared (Kenrick, 1997). The early forms of plants colonized on ancient land surfaces covered with cryptogamic soils, a microbiotic crust created by fungi, bacteria, and algae atop silts and clays (Prothero, 2013). The advent of the roots systems allowed a medium to transport water and other nutrients into the plant from the ground. Additionally, lignin evolution seen in tracheophytes provides the plant with enough structural support to not only grow to heights of 40m (such as the case with Lepidodendron), but to also allow effective water transport. Lignin also gives the plant durability and resilience to degradation, which improves preservation potential.
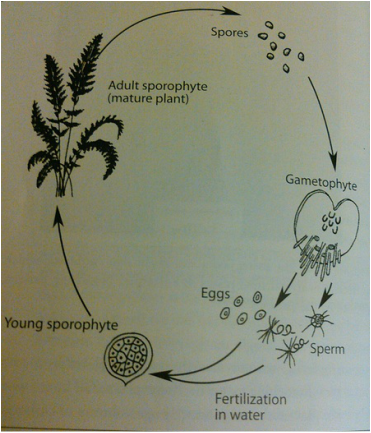
The terrestrial seedless vascular plant life cycle include a gametophyte and a diploid (sporophyte) phase, with the latter being the most prevalent in the fossil record (Figure 6). The sporophyte produces spores (through meiosis) which are picked up by the gametophyte. The gametophyte produces gametes in moist soil through mitosis, which leads to the fertilization of embryonic sporophytes that mature to macro sporophytes, and the cycle continues.
Lycophyte emergence in the fossil record occurs during the late Silurian. As a monophyletic group, both the lycophytes and the Zosterophyllophyta comprise a clade, with most evidence suggesting that the lycophytes originated from the zosterophylls (Taylor et al, 2009). One of the first lycophytes (genus Baragwanathia) is dated to be late Silurian, suggesting a possible pre-Devonian appearance of the group (Gensel, 2001). The lycophytes radiated in both diversity and size during the early Carboniferous, and then declined in the late Carboniferous.
Sphenophyte origins tend to be unclear when examining the fossil record. Typically considered to be derived from the trimerophytes, several plant species, such as Ibyka, exhibit characteristics similar to the sphenophytic branching system that pre-date true sphenophytes (Taylor et al, 2009). However, recent research suggests that sphenophytes and ferns could have descended from a common ancestor based on a TEM study (Transmission Electron Microscopy, a procedure that focuses electron beams to pass through a small, nanometers thick object that ultimately creates a higher resolution image for study) of Hyeniales (an organism containing both fern and sphenophyte features) and spores emitted by both sphenophytes and ferns that have similar structures (Grauyogel-Stamm, 2009). Most pre- Carboniferous fossil records of sphenophytes are sparse.
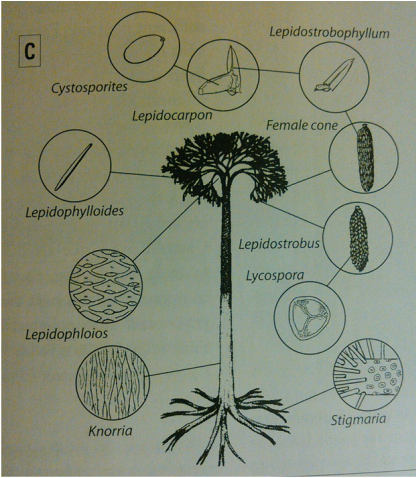
Plants, such as lycophytes and sphenophytes, generally have a low preservation potential. Cellulose is more easily weathered than calcitic shell, for example, and leaves and branches tend to be destroyed during the early stages of the taphonomic process. Plants with lignin provide some durability and are generally more resistive to degradation than cellulose. Due to the variability in fossilization and difficulty in correlating plant assemblages, parts of the plant can become separated during the burial process and are given their own taxonomic name when discovered. For example, the species Lepidodendron comprises of roots (Stigmaria), leaflets (Lepidophylloides), cones (Lepidostrobus), and other parts of the plant that are generally not found together (Figure 7). Carboniferous swamps provided a stagnant environment for plants to be fossilized more readily, typically through carbonization or permineralization. In Carboniferous coal-bearing strata, plant fossils are likely to appear. Some branches, trunks, and roots can also be preserved as molds and casts, yielding a three- dimensional fossil instead of a flat carbon film or impression.
Furthermore, coal balls in stratigraphy contain fossilized vegetation. Histosols were the dominant soil order of the Carboniferous coal swamp environment. In the past, peats were nutrient rich, which is seen through fossilized evidence of coal balls. Coal balls are calcareous nodules of compacted plants. Lycophytes and sphenophytes may have coerced a more basic soil, allowing the build-up of calcareous coal balls in rocks such as limestone (Retallack, 1990). Modern swampy soils tend to not contain coal balls due to the acid-rich environment that restricts calcium carbonates to form (Retallack, 1990).
Anatomy & Taxonomy
Many ancient lycophytes appear as “scale trees” that had characteristic rhythmic scar patterns on their bark that signify leaves (microphylls) that have fallen off. Lycophytes were lignin-rich trees that constantly shed its leaves like typical modern trees. Unlike today's trees that are attached to branches, these lycophytes grew leaves spiraled up along the trunk. Cones that housed spores hung between the leaf and the stem. The tree was grounded by a root system that reinforced and nourished it.
Many ancient lycophytes appear as “scale trees” that had characteristic rhythmic scar patterns on their bark that signify leaves (microphylls) that have fallen off. Lycophytes were lignin-rich trees that constantly shed its leaves like typical modern trees. Unlike today's trees that are attached to branches, these lycophytes grew leaves spiraled up along the trunk. Cones that housed spores hung between the leaf and the stem. The tree was grounded by a root system that reinforced and nourished it.
The sphenophytes, such as Calamites, grew through root systems and rhizoidal colonies. The main stalk was hollow, and is usually preserved as an internal pith cast representing the evenly incremented longitudinal ridges. As the stalk grew vertically, branched whorls of leaves would protrude from nodes spaced periodically up the stem. Different groups of sphenophytes produced a variety of radial leaf assemblages, such as Annularia or Sphenophylum. A round strobilus sat atop the stem and housed spores (Figure 8).
Taxonomy in paleobotany is challenging because specific plant parts are typically given their own taxonomic name without an association to an actual plant. Fossilized plant parts, such as leaves, roots, cones, etc are often fragmented from the original organism when they are discovered and described. For example, the root fossil Stigmaria could be associated to the lycophyte organism Lepidodendron or Sigillaria. In order to simplify the potentially extensive nature of plant identification, this guide will focus on fossil lycophyte and sphenophyte groups commonly seen in Pennsylvania.
The Lycophyta division consists of the four extinct orders:
- Protolepidodendrales (herbaceous lycophytes, including Baragwanathia and Protolepidendron ),
- Lepidodendrales (tree lycophytes, such as Lepidodendron and Sigillaria),
- Pleumeiales (consisting of one genus: the unbranched Pleuromeia), and
- Drepanophycales (oldest known lycophytes from the Devonian).
- Lycopodiales (club mosses, such as Lycopodium),
- Selaginellales (spike moss Selaginella with its many species), and
- Isoetales (quillworts like Isoetites and Isoetes).
The Carboniferous swamps consisted of primarily Lepidodendrales. Lepidodendron species exhibit an indicative diamond shaped bark pattern. The following is a list of common Lepidodendron assemblages with descriptions (Figure 7)(adopted from Milsom, 2013):
- Knorria – Trunk. Large leaf cushions
- Lepidophloios- Branches. Similar to trunk, displaying leaf cushions
- Lepidophylloides- Long, narrow, and needle-like leaves. The length and shape of the leaf depends on where the leaf grew: long ones hanging from branches, small and broad ones closer to the trunk
- Lepidostrobus- Cones (“male”) hanging between the stem and leaves. Solitary; not seen in clusters. Contained Lycopora spores
Appearing alongside Lepidodendron, Sigillaria were somewhat shorter than other lycophytes of its time. Their leaves were clustered on the ends of branches, but scars show that they also grew spiraled up along the trunk similarly to Lepidodenron’s leaf scars, but are more circular and not scaled in appearance. Also, their trunks were smoother with furrows that ran vertically up the tree. Sigillaria cones were clustered, unlike the Lepidodendron’s singular cones. Overall, the tree typically had distol branches near the top where leaves and cones developed.
Lycophytes also had root systems that varied little amongst species. Due to this ambiguity, a separate taxonomic name is needed. Stigmaria, the lycophyte rootstalks, were horizontal, simple, and moderately branched that not only housed rootlets to nourish the lycophyte, but also supported the towering aboveground portion. The fossil has characteristic small circular scars that originally housed the rootlets and appear somewhat evenly spaced.
The Sphenophyta division consists of the two extinct orders:
- Pseudoborniales (rare Devonian order with the single species Psuedobornia ursina) and
- Sphenopyllales (small plants found in the understories of Carboniferous forests).
The only extant order of the Sphenophyta is the Equisetales (horsetails). A major Carboniferous sphenophyte is the Calamites and its species. Usually preserved a pith cast, these equisetales are characterized by noticeable vertical furrows that are interrupted by small nodes where whorls of leaf branches would protrude. Annularia (Calamites foliage) appears in radiating whorls of leaflets. Sphenophyllum is another equisetale fossil commonly seen in Carboniferous strata.
Pennsylvania Examples & Fossil ID Guide
Pennsylvania stratigraphy, especially in Western PA, consists primarily of Carboniferous rock units. The Carboniferous can be broken down into the Mississippian (roughly 360-325 Mya) and Pennsylvanian (325-300 Mya) Periods (Figure 9). Pennsylvanian rock units contain fossils such as Lepidodendron and Calamites due to marsh environments during the time of deposition. The paleoecology in Pennsylvania during the Pennsylvanian Period slowly changed due to the rising sea level, providing an ideal habit for lycophytes and sphenophytes as originally arid lands become more susceptible to flooding. However, by the end of the Pennsylvanian, complexity in edaphic properties, tectonics, and geomorphic processes led to an overall drier and harsher environment for wetland thriving organisms (Feng, 1989).
Pennsylvania stratigraphy, especially in Western PA, consists primarily of Carboniferous rock units. The Carboniferous can be broken down into the Mississippian (roughly 360-325 Mya) and Pennsylvanian (325-300 Mya) Periods (Figure 9). Pennsylvanian rock units contain fossils such as Lepidodendron and Calamites due to marsh environments during the time of deposition. The paleoecology in Pennsylvania during the Pennsylvanian Period slowly changed due to the rising sea level, providing an ideal habit for lycophytes and sphenophytes as originally arid lands become more susceptible to flooding. However, by the end of the Pennsylvanian, complexity in edaphic properties, tectonics, and geomorphic processes led to an overall drier and harsher environment for wetland thriving organisms (Feng, 1989).
Pennsylvanian stratigraphy commonly contains lycophyte and sphenophyte fossils. Exposed outcrops, such as road cuts, pits, and quarries are the best places to find fossils. Plant fossils are most likely found in sedimentary rock (shales, mudstones, sandstones, limestones, etc) nearby coal units. Pelitic rocks, such as shales and siltstones, typically reflect a low energy, anoxic environment that improves plant preservation potential. Fossils are tougher to preserve in mature sandstones because they indicate a high energy marine environment, such as deltaic or tidal ecosystems, that tend to destroy plants during taphonomic processes and also can be too coarsely grained to sustain features.
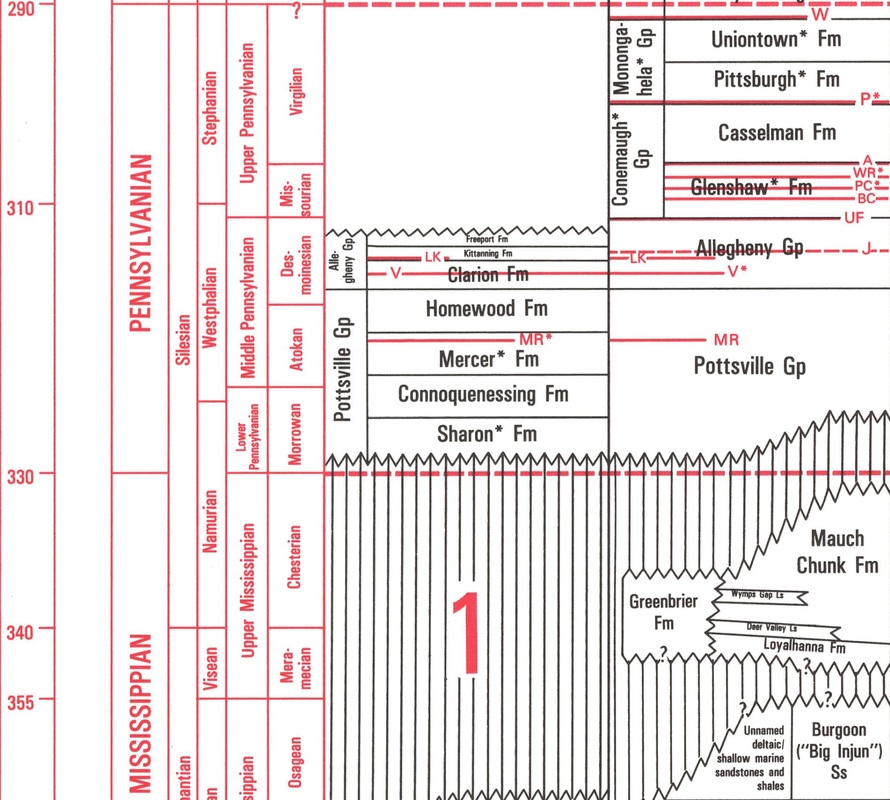
Figure 10. Stratigraphic column from the Pennsylvania Geological Survey showing Western Pennsylvania units during most of the Carboniferous Period. Mya shown to the left of the figure. Vertical bars indicate missing stratigraphy. Horizontal red lines through units represent important coal beds. Note: In Eastern PA, The Llewelyn Formation lies above the Pottsville Group/ Formation instead of the Allegheny, Conemaugh, and Monongahela. (Berg, et al, 1993)
The Western PA coal-containing Pottsville, Allegheny, Conemaugh, and Monongahela Groups (Figure 10) (Berg et al, 1993) often contain lycophyte and sphenophyte fossils. For example, many shales seen around coal units, such as the Pittsburgh and Waynesburg coal, will have many types of flora from wetland environments (Harper, 1990). Additionally, the Llewellyn Formation seen in Eastern Pennsylvania was deposited concurrently during the Pennsylvanian Period with fossilized lycophytes and sphenophytes found around anthracite layers (Oleksyshyn, 1982). The famous St. Clair pyrophyllite-coated plant fossil beds in Schuylkill Co, PA are part of the Llewellyn Formation. Found directly near the Buck Mountain coal bed, the Llewellyn Formation comprises many portions of lycophyte and sphenophyte flora including Sphenophyllum, Annularia, Asterophyllites, and Lepidodendron assemblages (Wagner, 1997). Units below the Pennsylvanian, such as the Mississippian Burgoon Sandstone Formation, rarely contain fossilized plants. However, In the Southern Anthracite Coal Fields, the uppermost portion of the Mauch Chunk (found stratigraphically above the Burgoon Sandstone) contains about five feet of shales that have well-preserved fossilized plants (Jennings, 1985).
The following lists are synthesized based off of USGS Fossil Flora survey of PA (Lesley, 1889) and Geology of PA (Rogers, 1858). Additional descriptions were adapted from plates from Lesquereux (1879):
Knorria – trunks of Lepidodendron. More elongate and spiraled leaf scars without strong rounded pattern. Large and discrete upwardly pointing scars (Figure 16 & 24).
- K. imbricata, acicularis
Ulodendron - Large round-oval branching scar rings, short leaves ( Figure 17).
- U. elongatum, minus
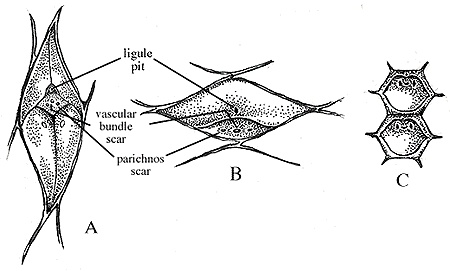
Lepidophloios – leaf cushion is broader than it is long. The parichnos and vascular bundle scar are grouped to the side of the overall scar (suggesting that the leaf scar is placed on the lower part of the cushions) with a central ligule and are typically “eye” shaped horizontally as compared to Lepidodendron, which has vertical eye-shaped leaf cushions (See Figure 11).
- L. laricinus, marcolepidotus
Lepidophyllum – bladed spore cases that may possibly be joined with Lepidostrobus (cones of Lepidodendron) (Figure 18). Difficult to describe in detail when not connected to original stems.
- L. acuminatum, affine, auriculatum, brevifolium, campbellianum, foliaceum, gracile, hastatum, lanceolatum, mansfieldi, obtusum, plicatum, proliferum, stantoni, undulatum
- L. acuminatum – long blade, more lanceolate
- L. brevifolium – short blade, curved at base (~7mm broad by 6mm long.)
- L. obstusum – longest and linear blades, rounded near top.
Lepidostrobus - cylindrical to oblong lycophyte strobili cone (Figure 18). Also comprised of parts such as Lepidophyllum (leaf spore cases) and Lepidostrobophyllum (sporophylls) (Figure 19). Lepidocarpon are also cones, but the sporangium is almost completely closed by sporophyll and represent an evolutionary peak for lycophytes (Andrews, 1961) (Figure 20).
Lycopodites – not too common in PA, small and herbaceous; branch and leaf fruit spikes (Figure 14).
- L. matthewi, simplex
Sigillaria – Scale trees that did not branch as much as Lepidodendron, overall a smaller and simpler arborescent form (Figure 21, 22, & 23). Typically either has furrows running vertically with circular/elliptical leaf scar bases between them, or hexagonal to rounded leaf scars arranged in a particular pattern with no furrows. Leaf scars tend to be smaller than that of Lepidodendron. Also had cones (Sigillaristrobis) and leaves (Sigillarioides). Separated into subgroups Eusigillaria and Favularia based on leaf base characteristics and bark surface (Taylor et al, 2009).
- S. alveolaria, acuminata, aculata, alternans, alveolaris, angusta, approximate, attenuata, conjugata, brardii, brochantii, brentonensis, brongniarti, brownie, catenulata, catenoides, davreuxi, defrancii, discoidea, dilatata, dubia, elongata, elegans, eliptica, eminens, fissa, flexusoa, grandeuryi, hexagona, lacoei, laurenciana, laveretti, lepidodendrifolia, lescurii, mammillaris, marginata, menardi, monostigma, obliqua, obovata, ovalis, pittsoniana, planicosta, polita, pulchra, pyriformis, rectostriata, reniformis, reticulata, saullii, schimperi, sculpta, scutellata, serlii, sillimani, simplicitas, rugosa, spinulosa, stellata, striata, suborbicularis, sydensis, tessellata, venosa, weissii, yardleyi
- S. monostigma – variable leaf scars (typically eye to coffin shaped) that run diagonally between bark wisps. Usually rounded and simple scars. No furrows
- S. brardii – exhibits a range of characteristics. Hexagonal to rounded- rhomboidal scar pattern with no furrows. One continuous pattern as seen in Lepidodendron
- S. fissa – distinct leaf scars, rounded mouth to “lip” shaped. Ligule abover center. No furrows, aligned vertically and evenly spaced.
- S. obliqua – similar to S. fissa, but bark pattern is more distorted and oblique. Somewhat central ligule, not as distinct, and leaf scars are not vertically aligned. Somewhat vertically ribbed
- S. dilata – rounded to lenticular scars that are close together and small. Usually flat with a medial vascular scar.
- S. stellata – leaf scars are rounded (broader than long), somewhat rhomboidal, has interesting striations that radiate from scar in star- like wrinkles
- S. tessellata –variable species. Usually small scars that are oval to hexagonal shaped that are strongly and tightly aligned vertically
- S. lescurii –narrow ribs. Furrows contain closely packed leaf scars that are large and ovate
- S. attenuata – ribbed similarly to S. lescurii, but leaf scars in furrows are spaced apart
- S. yardleyi – small, but distinct, leaf scars aligned vertically in narrow furrows. Smooth ribs
- S. sillimanni – narrow ribs, fairly close ovate scars that enlarge towards a rounded base
- S. ovalis – flattened ribs, furrows small. Simple scars
- S. obovata – broader ribs than most other species, small scars are spaced in deep and narrow furrows
- S. reniformis – furrows less distinct, reniform-kidney shaped scars
- S. elongata – ribs and furrows straight, deep, and distinct. Oblong scars
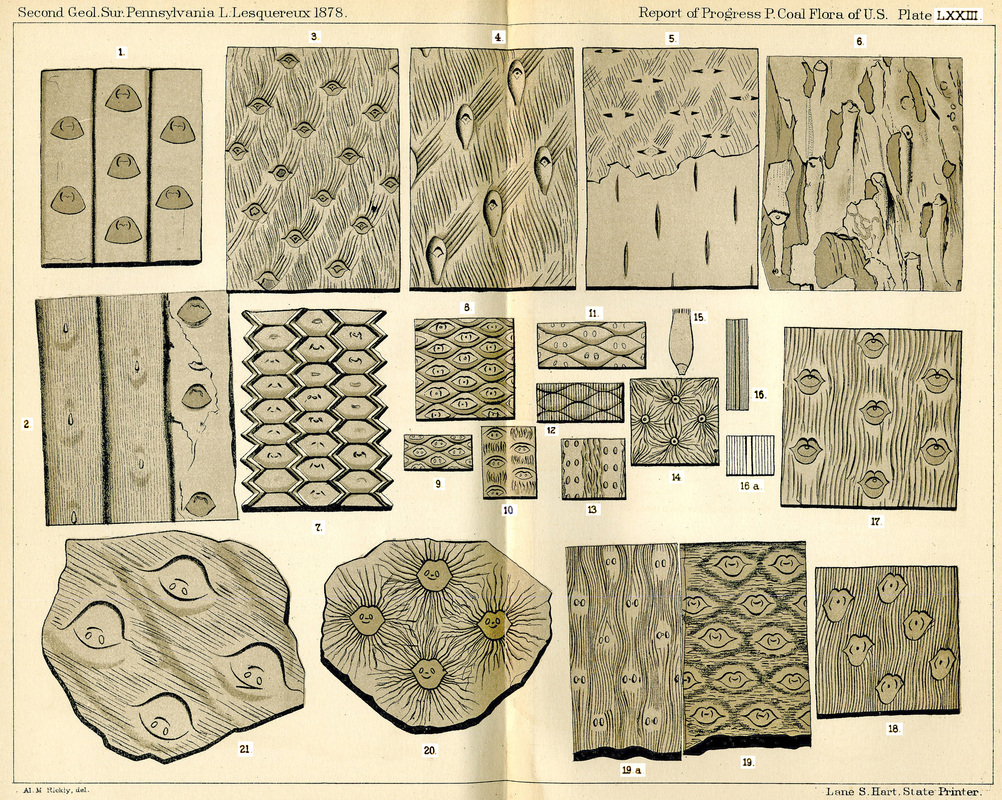
Figure 21. Sigillaria bark patterns. (1) Sigillaria polita. (2) Sigillaria Yardleyi. (3-6) Sigillaria monostigma. (7) Sigillaria ichthyolepsis. (8-16a) Sigillaria Brardii. (17) Sigillaria fissa. (18) Sigillaria obliqua. (19&19a) Sigillaria reticulata. (20) Sigillaria stellata. (21) Sigillaria Schimper. (Lesquereux, 1879) (http://www.georgesbasement.com/LesquereuxAtlasP/Lesquereux-Plate73.htm)

Figure 22. More Sigillaria bark species. (1) Sigillaria hexagona. (2-4b) Sigillaria tessellate. (5,6) Sigillaria mamillaris. (7,8) Sigillaria Lescurii. (9) Sigillaria attenuate. (10) Sigillaria Leptoderma. (11) Sigillaria Volzii. (12-12b) Sigillaria Lacoei. (Lesquereux, 1879) (http://www.georgesbasement.com/LesquereuxAtlasP/Lesquereux-Plate72.htm)
Stigmaria – roots of lycophytes that typically have uncertain origin unless directly found attached to the rest of the organism. Most are thick, sub cylindrical rounded casts with staggered ovate pits or node scars that represent rootlet attachment sites. Varieties based on symmetry, scar shape, and textures (Figure 24 & 25).
- costata, exigua, ficoides , irregularis, minuta, minutissima, perlata, perlata, pusilla, radicans, rugose, undulata

Figure 24. Varieties of Stigmaria species with examples of Sigillaria and Knorria. (1-4 & 6) Stigmaria ficoides varieties. (5&7) Stigmaria stellaris. (8) Stigmaria umbonata. (9) Stigmaria Halonia tuberculata ?. (10-10b) Sigillaria Owenii. (11) Stigmaria in situ. (12-13) Stigmaria sp. (14&15) Knorria imbricata. (Lesquereux, 1879) (http://www.georgesbasement.com/LesquereuxAtlasP/Lesquereux-Plate74.htm)
Sphenophyta ID:
Order Sphenophyllales
Order Sphenophyllales
- Usually represented by the genus Sphenophyllum, a small herbaceous plant that lived during the late Devonian to Permian (Figure 26 and 29). All “Sphenophyllum” species tend to have 6 to 10 leaf “wedges” per whorl with simple stems and have morphological variability based on whorl symmetry and vein characteristics, making species level identification occasionally difficult. Many Sphenophyllum species are synonymous with one another due to a misinterpreted taxonomic history. “Sphenophyllum” was originally used to describe solely a leaf arrangement, but now the term is connected with roots, stems, leaves, and even whole plants (Taylor et al, 2009). To avoid confusion, this guide will focus on foliage due to established research in leaves.
- S. bifurcatum, cuneiform, emarginatum, erosum, fontainianum, longifolium, oblongifolium, primaevum, schlotheimi, tenerimum, trifoliatum, vetustum
- S. emarginatum – fanned, narrows towards stem attachment in well-preserved specimens, rounded wedge. Longer and narrower leaves.
Order Equisetales
- Calamitacae
Calamites – Late Mississippian to Permian. Interestingly, the plant could range from 30 to 60 feet tall, and was typically around only 3 to 6 inches in diameter (Rogers, 1858), but could be up to 2 feet diameter (Taylor et al, 2009). They usually grow from underground lateral root growths (rhizomal). Many Calamites fossils are steinkerns (hollow portion of the original organism’s canal become infilled with sediment during taphonomic processes and then became fossilized as the outer vascular tissue becomes broken down), also known as pith casts. They are characterized by cylindrical stems with vertical furrows separated by nodes. Roots and branches are rarely attached with stem (Figure 27 & 28).
- C. approximates, bistriatus, cannaeformis, cistii, cruciatus, decoratus, disjunctus, dubius, pachyderma, ramifer (ramosus), suckowii, undulates
- C. approximatus – closely spaced lateral nodes as compared to other species. Indistinct rib marks, thick cortex. Articulations seem tightened/contracted. Deep furrows that sometimes converge at articulation spots
- C. suckowii – common Calamites. Ribs somewhat rounded zig-zag appearance along articulation running horizontally in well preserved specimens. Nodes and scars visible
- C. ramifer (ramosus) - very distinct horizontal nodes and articulations. Ribs are flat and furrows are narrow
- C. cannaeformis – variable articulation lengths, somewhat large ribbed compared to other species, rounded convex ribs, distinct leaf scars
- C. cistii – equi-distant articulations, narrow ribs, convex/half round. Usually smaller than other species
- C. dubuis & C. bistriatus – very similar species, long stems and internodes, distinct articulations and furrows, narrow ribs
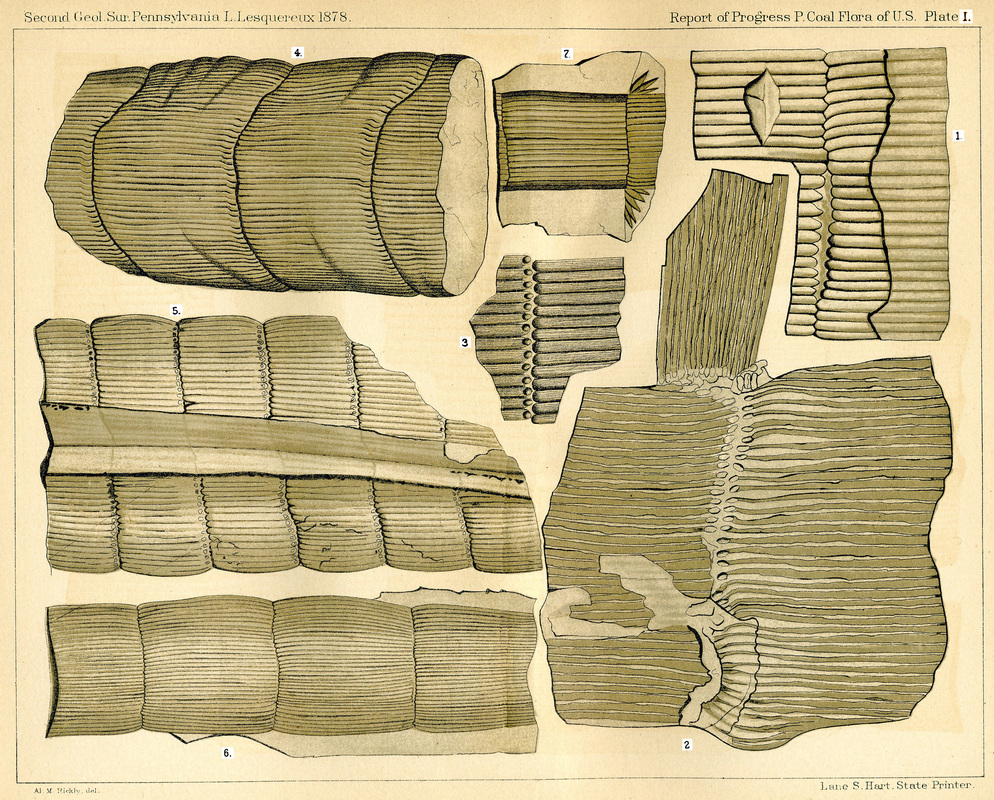
Figure 27. Calamites examples and Bornia (another sphenophyte). (1) Calamites cannaeformis. (2) Calamites ramosus. (3&4) Calamites Suckowi. (5) Calamites approximates. (6) Calamites Cistii. (7) Bornia radiata. (Lesquereux, 1878) (http://www.georgesbasement.com/LesquereuxAtlasP/Lesquereux-Plate01.htm)
Foliage: Three main groups (Figure 29). Species names are differentiated based on leaf shape and arrangements. Important to note that during fossilization, leaflets will compress onto one plane with the stem, so each leaf may not appear to be of similar size. Descriptions adapted from Walton (1936):
Lobatannularia – (obscure Permian genus, not typically seen in PA strata)
Lobatannularia – (obscure Permian genus, not typically seen in PA strata)
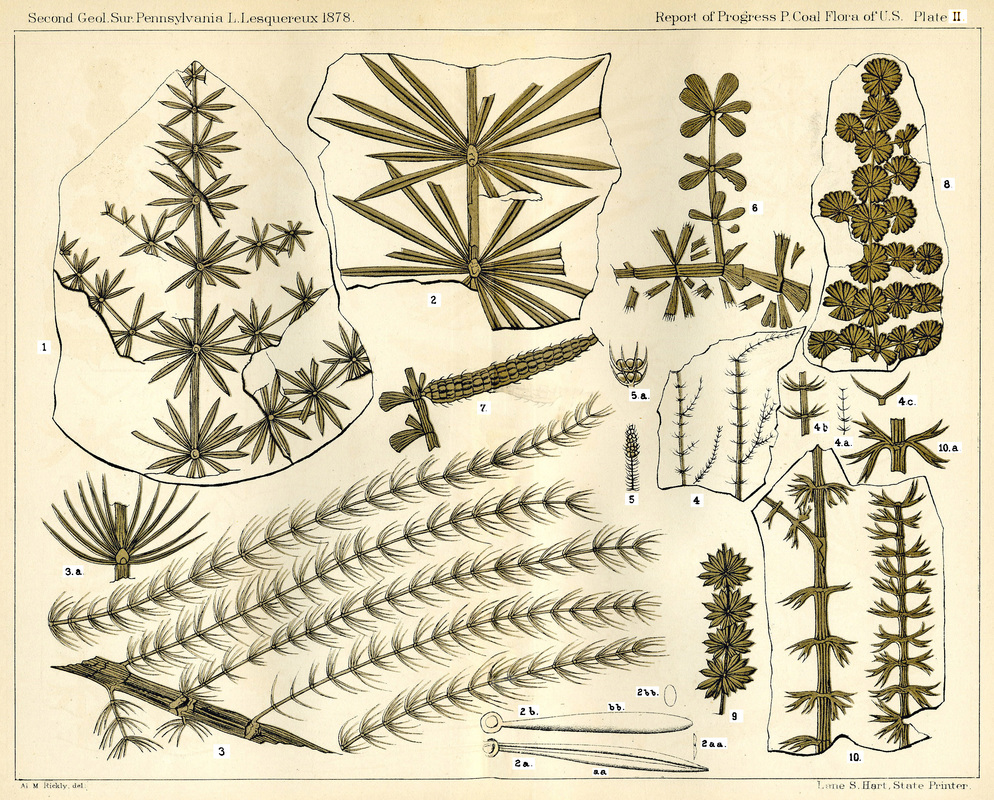
Figure 29. Sphenophyte foliage. 1-2aaa) Annularia longifolia. 2b & 2bb) Annularia inflata. 3&3a) Asterophyllites equisetiformis. 4-5a) Asterophyllites gracilis. 6-7) Sphenophyllum Schlotheimii. 8&9) Annularia sphenophylloides. 10&10a) Sphenophyllum bifurcatum. (Lesquereux, 1879) (http://www.georgesbasement.com/LesquereuxAtlasP/Lesquereux-Plate02.htm)
Annularia – most common leaf arrangement for Calamites fossil. Typically slender to tear drop shaped leaves in a disk-like arrangement (Figure 29 & 30).
- A. fertilis, galioides, longifolia, radiata, roemingeri, sphenophylloides, spinulosa, stellate, hoskinsii (Taylor et al, 2009)
- A. sphenophylloides – common Annularia with whorls of 12-18 spatulate leaves, each 3 to 10mm long and 1-3mm wide. Tend to be convex/concave shape. Terminal expansion of central vein (appears tapered when examining the leaf from tip to stem, in other words, each leaf thins out towards the stem attachment). Somewhat ovular to feather-outline in shape
- A. galioides – similar to A. sphenophylloides, but is slightly more slender and lenticular in leaf shape. Typically 8 leafs per whorl (tends to be less clustered than A. sphenophylloides)
- A. radiata – the most slender and lanceolate shaped leaves. Does not exhibit “swelled” leaves, but slightly spatulate
- A. stellata – no terminal expansion of vein, which is seen in A. sphenophylloides. Slightly different forms, all slender and clustered per whorl, typically more so than A. radiata. (similar appearance to that of A. fetilis and A. spinulosa)
- A. longifolia – 18-24 leaves per whorl. (plane propeller shape. Elongate and rounded)
Asterophyllites – Characterized by thin stems and a more parallel offshoot, yielding a preferred orientation of leaves pointing towards the end of the branch. (Figure 29). Generally long and stiffer leaflets that point upward in the same direction (as compared to Annularia, which tends to have no preferred orientation when compressed and is usually more radial).
- A. apertus, anrthracinus, crassicaulis, equisetiformis (erectifolius), fasciculatus, foliosus, gracilis, longifolius, ovalis, sphenophylloides, sublaevis
- A. longifolius – extremely long and slender (almost needle-like), upwardly pointing leaflets. Radial offshoot in whorls
- A. equisetiformis – upwardly pointing like longifolius, but with thicker leaflets that have superficial appearance of Annularia stellata. linear leaves
References Cited
- Andrews, H., 1961, Studies in Paleobotany: John Wiley & Sons, Inc.
- Berg, T. M., McInerney M. K., Way, J. H., MacLachlan, D. B., 1993, Stratigraphic correlation chart of Pennsylvania: Commonwealth of Pennsylvania, Department of Environmental Resources.
- Cobb, B., Farnsworth, E., & Lowe, C., 2005, A field guide to ferns and their related families: northeastern and central North America: Houghton Mifflin Harcourt, v.2
- DiMichele, W., 1985, Diaphorodendron, gen. nov., a segregate from Lepidodendron (Pennsylvanian Age): Systematic Botany, American Society of Plant Taxonomists, v. 10, p. 453-458.
- Feng, B. C., 1989, Paleoecology of an upper Middle Pennsylvanian coal swamp from western Pennsylvania, USA.: Review of palaeobotany and palynology, v. 57(3), p. 299-312.
- Gensel, P. G., & Berry, C. M., 2001, Early lycophyte evolution: American Fern Journal, v. 91(3), p. 74–98.
- Grauvogel-Stamm, L., Lugardon, B., & Wellman, C. H., 2009, Phylogeny and evolution of the horsetails; evidence from spore wall ultrastructure: Review of Paleobotany and Palynology, 156(1-2), p.116-129.
- Harper, J. A., 1990, Fossil Collecting in the Pittsburgh Area: Pittsburgh Geological Society Guidebook, p. 50
- Jennings, J. R., 1985, Fossil plants from the Mauch Chunk Formation of Pennsylvania: morphology of Adiantites antiquus: Journal of Paleontology, p. 1146-1157.
- Kenrick, P., & Crane, P. R., 1997, The origin and early evolution of plants on land. (Cover story): Nature, v.33, p.389(6646).
- Lesley, J., 1889, A dictionary of the fossils of Pennsylvania and neighboring states named in the reports and catalogues of the Survey: Board of commissioners for the geological survey. v.1
- Lesquereux, L., 1879, Second Geological Survey of Pennsylvania: report of progress p. description of the coal flora of Pennsylvania and of the Carboniferous formation throughout the United States: http://www.georgesbasement.com. (Accessed April 2016)
- Milsom, C., & Rigby, S., 2013, Fossils at a glance: John Wiley & Sons.
- Oleksyshyn, J., 1982, Fossil plants from the anthracite coal fields of eastern Pennsylvania: Pennsylvania Geological Survey.
- Phillips, T. L., 1980, Stratigraphic and geographic occurrences of permineralized coal-swamp plants—Upper Carboniferous of North America and Europe: Biostratigraphy of fossil plants: Dowden, Hutchinson and Ross Stroudsburg, PA, p. 25-92.
- Prothero, D. R., 2013, Bringing fossils to life: An introduction to paleobiology: Columbia University Press.
- Purves et al., 1994, Life: the science of biology, 4th edition: Sinauer Associates
- Retallack, G.J, 1990, Soils of the Past: Unwin-Hyman, London
- Rogers, H. D., 1858, The geology of Pennsylvania: a government survey: W. Blackwood and sons. v.2
- Sacks, O., 2011, Field trip: hunting horsetails: The New Yorker.
- Stewart WN, Rothwell GW., 1993, Paleobotany and the evolution of plants: New York, NY, USA: Cambridge University Press.
- Taylor, E. L., Taylor, T. N., & Krings, M., 2009, Paleobotany: the biology and evolution of fossil plants: Academic Press.
- Wagner, R. H., & Lyons, P. C., 1997, A critical analysis of the higher Pennsylvanian megafloras of the Appalachian region: Review of Palaeobotany and Palynology, v.95(1), p.255-283.
- Walton, J., 1936, On the factors which influence the external form of fossil plants; with descriptions of the foliage of some species of the Palaeozoic equisetalean genus Annularia Sternberg: Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences, v. 226(535), p.219-237.